High-calorie diets uncouple hypothalamic oxytocin neurons from a gut-to-brain satiation pathway via $ąppa$-opioid signaling
Abstract
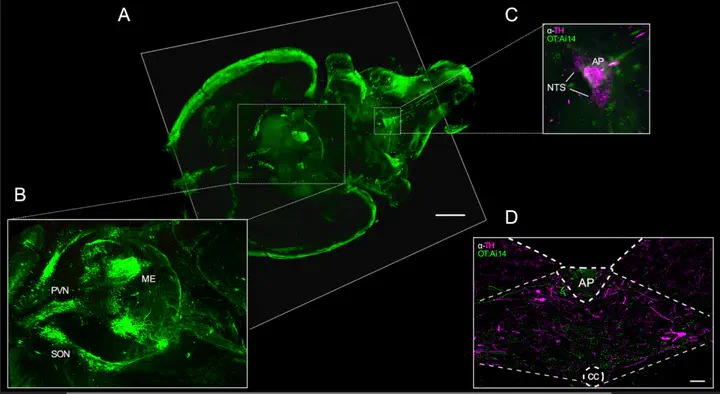
Oxytocin-expressing paraventricular hypothalamic neurons (PVN^OT neurons) integrate afferent signals from the gut, including cholecystokinin (CCK), to adjust whole-body energy homeostasis. However, the molecular underpinnings by which PVN^OT neurons orchestrate gut-to-brain feeding control remain unclear. Here, we show that mice undergoing selective ablation of PVN^OT neurons fail to reduce food intake in response to CCK and develop hyperphagic obesity on a chow diet. Notably, exposing wild-type mice to a high-fat/high-sugar (HFHS) diet recapitulates this insensitivity toward CCK, which is linked to diet-induced transcriptional and electrophysiological aberrations specifically in PVN^OT neurons. Restoring OT pathways in diet-induced obese (DIO) mice via chemogenetics or polypharmacology sufficiently re-establishes CCK’s anorexigenic effects. Last, by single-cell profiling, we identify a specialized PVN^OT neuronal subpopulation with increased κ-opioid signaling under an HFHS diet, which restrains their CCK-evoked activation. In sum, we document a (patho)mechanism by which PVN^OT signaling uncouples a gut-brain satiation pathway under obesogenic conditions.